 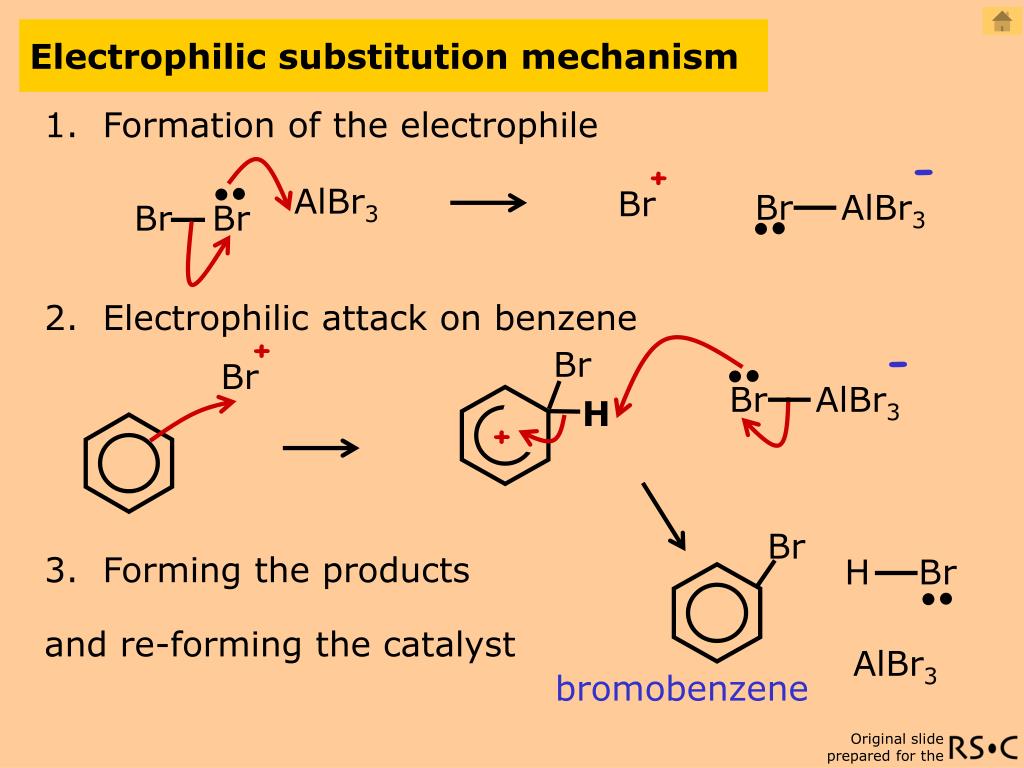
What bonds formed and what bonds broke in this reaction? Instead, if you treat benzene with Cl 2 you will eventually (and very slowly) obtain the following product: chlorobenzene. That’s actually not the product that we observe! Our first guess might be that benzene reacts with Cl 2 to give an “addition” product like that shown below (albeit more slowly than with a “normal” alkene): Knowing this, how might we expect electrophiles like Cl 2 or Br 2 to react with aromatic compounds like benzene? 2. high temperatures, high pressures of H 2 , extended reaction times ) in order to successfully add H 2 to benzene, relative to “typical” alkenes. Hydrogen (H 2) can be made to add across most alkenes in the presence of a catalytic amount of finely divided palladium on carbon ( Pd/C), but you really need to break out the can of Aldrich Brand Whup-Ass ® (i.e. You might also recall that benzene itself is unusually difficult to hydrogenate. We’ve seen that benzene has unusual stability (36 kcal/mol of resonance energy) relative to what we’d expect for theoretical “cyclohexatriene”, which would certainly lead us to predict that the pi bonds in aromatic molecules will be less reactive, relative to alkenes. Since we’re on the topic of benzene, it’s natural to wonder how well the pi bonds in aromatic systems (like benzene) compare in reactivity to the pi bonds in alkenes, and by extension, how the reactions of aromatic compounds compare to the reactions of alkenes. Chlorination of alkenes with Cl 2 is a classic example: That’s where we break a (relatively weak) C-C (pi) bond and form two new single bonds to carbon. When we covered the reactions of alkenes a while back – a lot of reactions! – we saw that the vast majority fell into the class of reactions we call addition reactions. Alkenes Give “Addition” Products Upon Reaction With Electrophiles. Summary: The Six Key Electrophilic Aromatic Substitution Reactionsġ.Friedel-Crafts Alkylation and Acylation.The Key Pattern For Six Important Electrophilic Aromatic Substitution Reactions.Lewis Acids Accelerate The Rate of Electrophilic Aromatic Substitution Reactions.  
Alkenes Give “Addition” Products Upon Reaction With Electrophiles.Each of these reactions requires an acid catalyst to activate it so that the relatively unreactive aromatic ring will attack it.There are six key electrophilic aromatic substitution reactions in most introductory organic chemistry courses: chlorination, bromination, nitration, sulfonation, Friedel-Crafts alkylation, and Friedel-Crafts acylation.In electrophilic aromatic substitution a C-H bond is broken and a new C-E bond (E being an electrophilic atom such as Cl, Br, N…) is formed.The Six Key Electrophilic Aromatic Substitution Reactions 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
